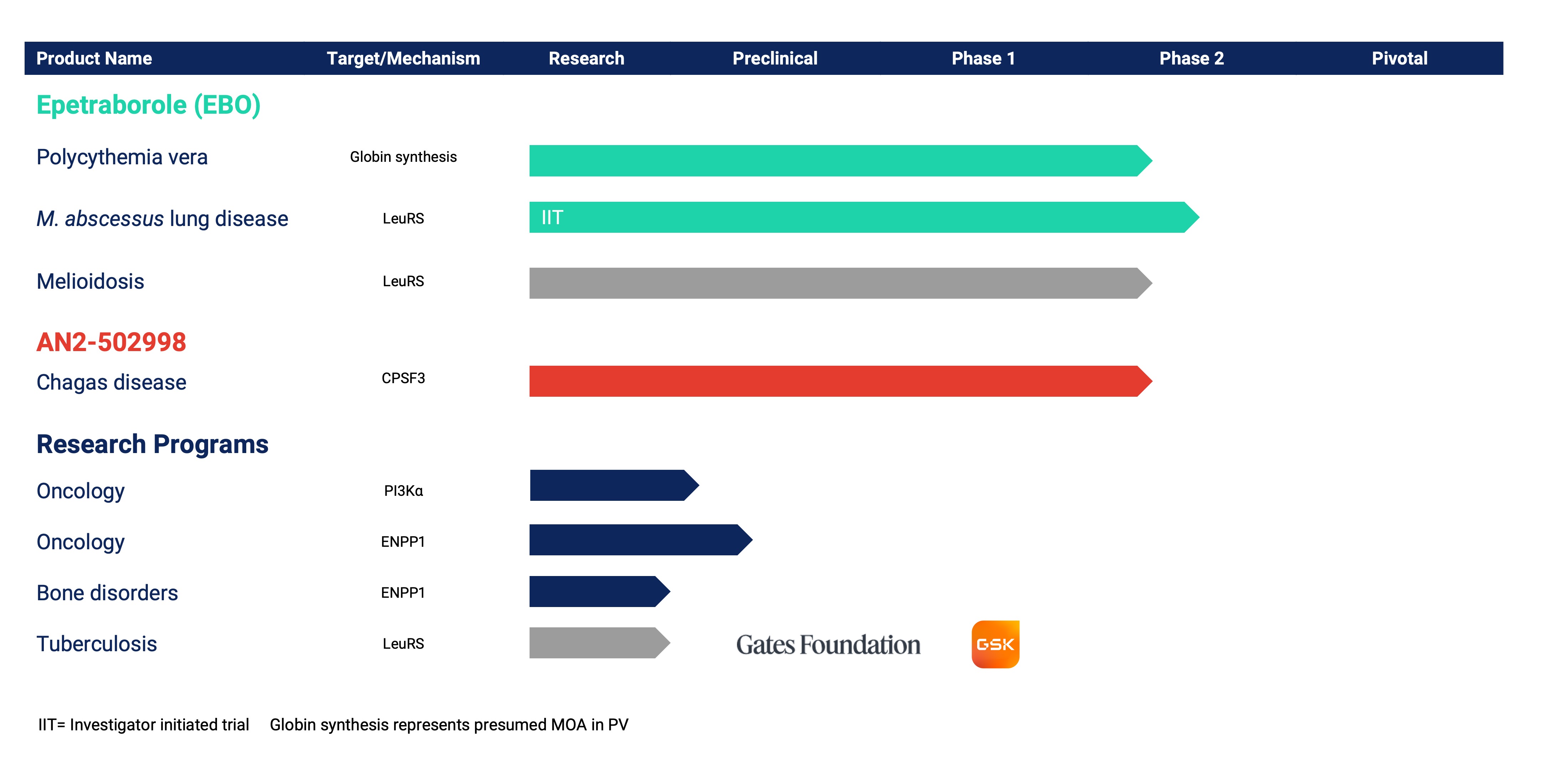
Epetraborole
Polycythemia Vera
We are expanding the development of oral epetraborole through a Phase 2 proof-of-concept clinical study in adults with phlebotomy-dependent polycythemia vera (PV). PV is a blood cancer characterized by overproduction of red blood cells in the bone marrow. This overproduction increases hematocrit, which can lead to serious medical complications including arterial and venous thromboembolic events. If untreated, PV can be life-threatening. Despite available therapies, many patients experience uncontrolled hematocrit levels and persistent symptom burden, requiring long-term management to maintain adequate disease control. PV is estimated to affect approximately 155,000 people in the U.S.
Mycobacterium abscessus complex lung disease
The U.S. Food and Drug Administration has cleared an Investigational New Drug Application to proceed with a Phase 2 investigator-initiated study in collaboration with Oregon Health and Sciences University (OSHU) evaluating epetraborole for the treatment of Mycobacterium abscessus (M. abscessus) complex lung disease. This multicenter, randomized, double-blind, placebo-controlled, prospective clinical study will be led by Dr. Kevin Winthrop, Professor of Public Health and Infectious Diseases at OHSU, in conjunction with other investigators across an estimated 10-15 sites in the U.S. M. abscessus lung disease is a serious and difficult-to-treat non-tuberculous mycobacterial infection requiring prolonged therapy including IV-only antibiotics and characterized by limited treatment options and high rates of morbidity, and 5-year mortality. No FDA-approved drugs currently exist for its treatment.